Last week, the New York Times published a rapturous profile of a drug marketing company called Medvi, which the paper declared to be the first one-ish man venture on track to surpass one billion dollars in revenue while leaning almost exclusively on AI to build and scale the business. The news was immediately met with a wave of online backlash over both the framing of the NYT piece and the ethics of the company at the center of it — and the company’s official response raises more questions than it answers.
Medvi is a company that Futurism has been aware of for some time now. It effectively operates as a marketing wrapper for telehealth companies that prescribe and fulfill orders for GLP-1 agonists and other compounded drugs — and as we first reported all the way back in May 2025, Medvi has engaged in a range of dishonest marketing practices. It’s plastered its website with AI-deepfaked images of phony patients passed off as the real deal, flaunted media logos insinuating that it garnered editorial coverage from mainstream media outlets when it hadn’t, and featured the name and likeness of at least one medical practitioner who told us that in reality he had nothing to do with the company.
The NYT‘s profile either downplayed or omitted all of these glaring ethical red flags. The article also definitively stated that Medvi had ceased its practice of using AI-fabricated before-and-after client photos — a declaration that, as we pointed out in a follow-up, wasn’t true.
We weren’t the only ones with qualms. As folks from across the health, tech, and business worlds were quick to point out online, at the time the NYT article was published, Medvi was being marketed on social media by a trove of Meta accounts featuring clearly fake doctors. In fact, in February 2026, Medvi LLC was issued a warning from the Food and Drug Administration (FDA) pointing to multiple violations on a Medvi-branded site, Medvi.io. In that February warning, the FDA said that the site, Medvi.io, was misleading consumers by featuring images of Medvi-branded pill bottles and vials of injectables — Medvi isn’t actually the compounder of any drugs it sells, leaning instead on various pharmacies — and suggesting that certain drug compounds Medvi peddles are FDA-approved when they aren’t. Both the fake doctors on social media and the FDA warning went unmentioned in the NYT piece.
Yesterday, in response to public backlash and new reporting from outlets including Futurism, Techdirt, Business Insider, and the pharma-focused outlet Drug Discovery & Development, Medvi and its founder, 41-year-old Matthew Gallagher, issued a statement that it characterized as a “response to external speculation” about the company and its marketing practices.
The statement Medvi provided focuses heavily on the context surrounding the FDA warning, noting that “in September 2025 and February 2026, the FDA sent an unprecedented number of warning letters to dozens of telehealth companies, drug companies and pharmacies regarding their direct-to-consumer advertising practices.” This is true: as we noted in our previous story, Medvi was one of numerous companies issued stark warnings amid a broader crackdown on the unruly GLP-1 marketplace.
But while the FDA letter is addressed to Medvi LLC, Medvi is insisting that it was an affiliate — basically, another marketer that funnels patients to Medvi in exchange for a financial kickback — that was technically in violation of the FDA’s warning.
“In one of the FDA’s letters, the URL mentioned is medvi.io — not MEDVi’s actual address of medvi.org,” reads the statement, which is attributed to Gallagher. “The letter addressed to MEDVi was directed at an affiliate marketing agency whose website contained outdated copy. We immediately reached out to the affiliate and required them to remove the materials allegedly at issue. We understand the affiliate also directly responded to the FDA.”
“My company MEDVi has never received a letter from the FDA,” the statement continues. “If we were to receive such a communication from any regulatory authority, we would act swiftly and collaboratively to address the matter.”
In response, we asked whether Medvi LLC — the company the FDA letter was addressed to — is Gallagher’s company that was featured in the NYT. We haven’t received any response.
When we first reported on the stolen before-and-after images on Medvi.io, the site had no affiliate disclaimers, and directed us to contact [email protected] with inquiries — the email that Medvi.org, which Medvi insists is its primary domain, still directs site-goers to today.
We asked Medvi when Medvi.io changed hands to an affiliate, and who this affiliate was, which also went unanswered..
Further, at the time the letter said Medvi.io was in violation of FDA guidelines — December 2025 — Medvi.org’s homepage featured the same AI-generated Medvi-branded bottles that the FDA warning said were “false or misleading” to consumers, as they suggested that Medvi is the compounder of the drugs it sells when it isn’t. According to archived versions of the site, Medvi.org appeared to have taken these images down after the FDA warning in February.
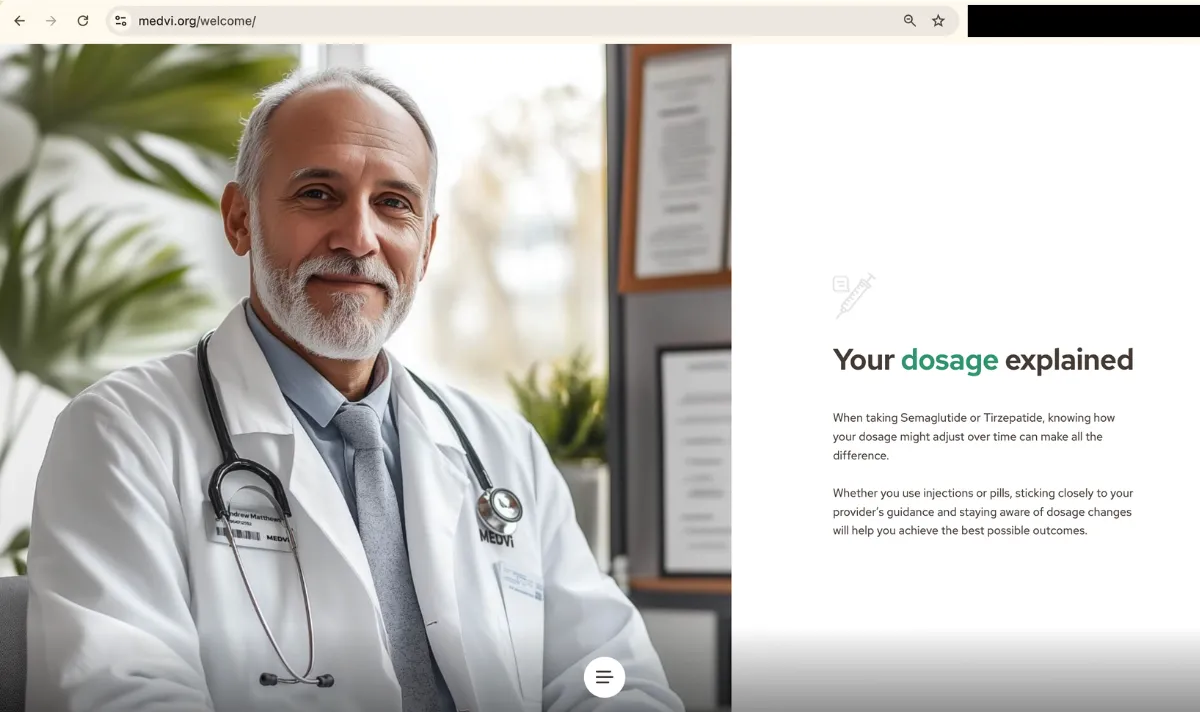
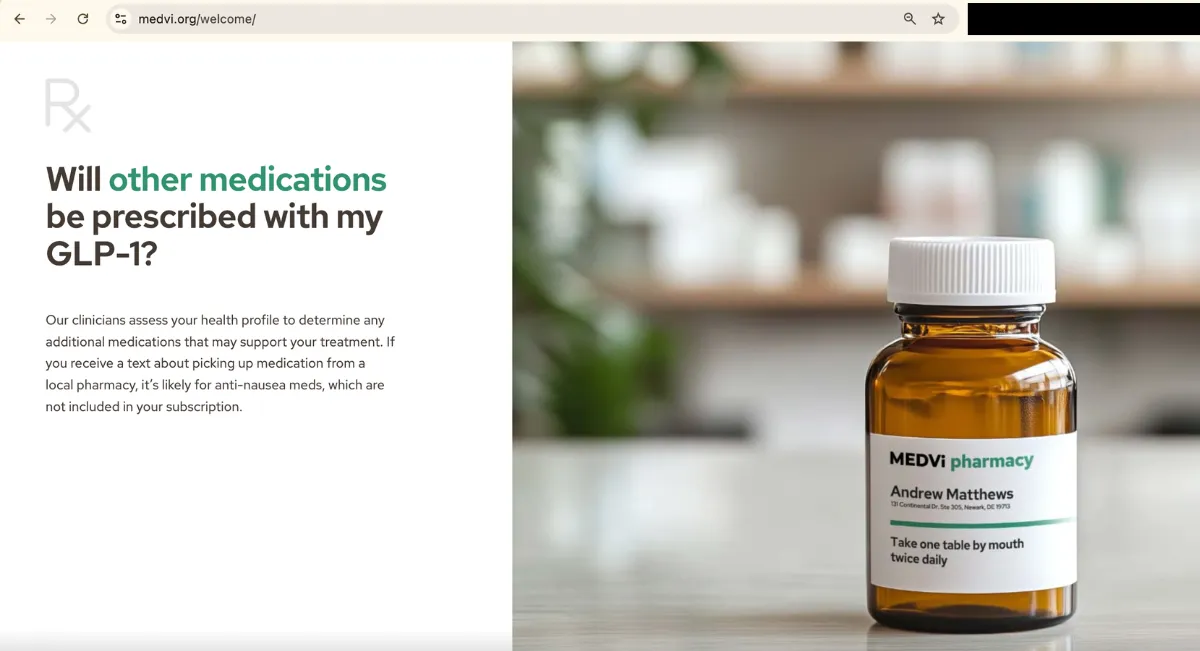
And yet, as of publishing this article, a functioning “welcome” page hosted by Medvi.org continues to prominently feature images of Medvi-labeled drugs — including a Medvi-branded bottle of oral tirzepatide tablets. Words on the bottle describe the tablets as “compounded medication” for “medical weight management.”
But oral tirzepatide isn’t FDA-approved for weight loss. In fact, while injectable tirzepatide is FDA-approved — tirzepatide is the active ingredient found in Eli Lilly’s Zepbound shot — the tablet version of the drug has no proven benefits. There’s no evidence that it’ll actually help buyers lose weight.
“Why don’t I just compound a little piece of cardboard and give it to you?” Dr. Jon Slotkin, a neurosurgeon and hospital executive at a large hospital system in Pennsylvania, told Futurism of the dubious compound. “There is exactly as much human data published that oral tirzepatide will help you lose weight as there is published data that compounded cardboard in a little tablet will help you lose weight. They’re identical in their amount of evidence.”
The same page also features images of AI-generated doctors in Medvi-branded lab coats. A label of one bottle reads “Medvi Pharmacy” and features instructions urging patients to “take one table [sic] by mouth twice daily.”
The page directs to a Medvi patient portal, and there are no affiliate caveats or disclaimers about FDA approval.
We asked Medvi whether Medvi.org was in violation of FDA guidelines at the time the warning was sent, and to clarify whether the operational “welcome” page we found was operated by Medvi LLC and in compliance with FDA rules. Again, we haven’t received any answer.
Elsewhere, Medvi’s statement addresses the controversy surrounding what the company describes as “‘so-called’ fake doctors,” claiming that it only “recently become aware of what appear to be advertisements featuring potentially AI-generated medical practitioners.” (Many of these accounts were deleted or altered almost immediately following inquiries from journalists.)
“Since we became aware of this issue, we have updated our marketing practices to make clear that this type of advertising and / or promotion is prohibited,” the statement continued. “We continue to proactively address this issue.” (“Proactively” means addressing something before it becomes an issue, not after you’re called out for it.)
Medvi’s response also failed to address the venture’s continued use of AI-generated before-and-after patient pictures. Asked whether it believes the use of fake, AI-generated patients is ethical, Medvi didn’t respond.
Still, the statement went on to say that Medvi is committed to “operating transparently.”
“Building a company the size of MEDVi and scaling so quickly involves many learning moments. At each of these stages, I have course-corrected immediately and appropriately. I will continue to do so,” Gallagher said in the statement. “The New York Times was recently given unprecedented access to MEDVi’s business information and also interviewed our business partners and other collaborators.”
“As I continue helping our customers achieve their health goals,” he added, “I remain committed to building and operating transparently.”
The post AI-Powered Drug Marketer Medvi Responds After Allegations About Fake Doctors and Patients appeared first on Futurism.




